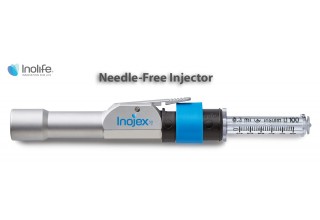
Inolife Announces the Commencement of Its Public Listing Process to Meet Growing Demand for Its Needle-Free Injection Technologies.

TORONTO, March 27, 2018 (Newswire.com) - Inolife R&D (“Inolife”), is pleased to announce today it aims for the Public Listing of its stocks on the Canadian Securities Exchange (CSE): currently private, the company commenced the listing process for trade on the CSE, by way of a Reverse Takeover (RTO) as a catalyst for accessing the capital needed to support its growth plan.
To that end, following the completion of its RTO all filings were made with a view to obtaining the timely listing of its stocks on the CSE. The application is subject to review and approval by the CSE, a Canadian trading platform aimed at venture market companies. The management team is confident that the application process will go smoothly and expects to be listed in Q2 2018.
This listing will provide enough working capital to support Inolife's growth plan and propel the Company into an exciting and rapidly growing industry. The ready-to-market products and the next generation needle-free injection technologies of Inolife will shape the Company into an industry leader.
Michael G. Wright, President & CEO
Management Commentary
Michael Wright, President and Chief Executive Officer of Inolife comments, “This listing will provide access to additional capital to support Inolife’s growth plan and propel the Company into an exciting and rapidly growing industry”. “The ready-to-market products and the next generation needle-free injection technologies of Inolife will shape the Company into an industry leader”, further added Mr. Wright.
About Inolife R&D Inc.
Inolife R&D Inc. is an emerging specialty medical device company focused on developing and commercializing novel drug delivery technologies. Inolife commercializes patented and FDA-cleared needle-free injection devices. The company was founded to take advantage of novel techniques of liquid jet and ballistics-based transdermal drug injection that improve patients’ quality of life by making medicines easier to administer, work better, and remove the anxiety and inconvenience associated with hypodermic needle injections.
For more information about Inolife and its products please visit www.inolifesciences.com.
FORWARD-LOOKING STATEMENTS
Certain statements contained herein are “forward-looking” and are based on the opinions and estimates of management, or on opinions and estimates provided to and accepted by management. Forward-looking statements are subject to a variety of risks and uncertainties and other factors that could cause actual events or results to differ materially from those expressed or implied. Readers are therefore cautioned not to place reliance on any forward-looking statement.
Contact:
Michael Wright
President & CEO, Inolife R&D Inc.
Direct: +1 (866) 834-3777
Email: mwright@inolifesciences.com
Source: Inolife R&D
Share:
Categories: Venture Funding, Financial News
Tags: cannabis, diabetes, drug, Inolife, medical device, needle-free, pharma, public listing, syringe